Backed by Clinical Evidence and Real-World Outcomes. Used in more than 2,000 hospitals across the U.S., join the clinicians who trust JADA® for fast, effective PPH management.
The JADA® System is an intrauterine device indicated to provide control and treatment of abnormal postpartum uterine bleeding or hemorrhage when conservative management is warranted. The JADA System uses low-level vacuum to stimulate physiologic uterine contraction and control bleeding. Supported by clinical evidence demonstrating rapid bleeding control, high effectiveness, and a favorable safety profile, JADA is designed to be easy to use and help clinicians respond quickly in the management of postpartum hemorrhage.
Rapid bleeding control, high effectiveness, and a favorable safety profile, the JADA System is designed to be easy to use and help clinicians respond quickly in the management of postpartum hemorrhage.
The JADA System can provide visible and observable control of bleeding within minutes.* Low-level vacuum (80 mm Hg +/- 10 mm Hg, not to exceed 90 mm Hg) induces physiologic contraction of the uterus to control and treat abnormal postpartum uterine bleeding or hemorrhage.1
In a clinical study (n=107), contraction of the uterus was observed in a median of 1 minute with control of bleeding observed in a median of 3 minutes.1

The uterus is composed of a web-like network of muscle fibers in its middle layer, called the myometrium. When the placenta separates from the uterine wall after childbirth, that portion of the myometrium is left with open blood vessels, resulting in bleeding. Typically, the uterus contracts and compression of those open vessels stops bleeding. In some instances, the uterine muscle fibers do not contract, causing uncontrolled bleeding, referred to as abnormal postpartum uterine bleeding or postpartum hemorrhage.
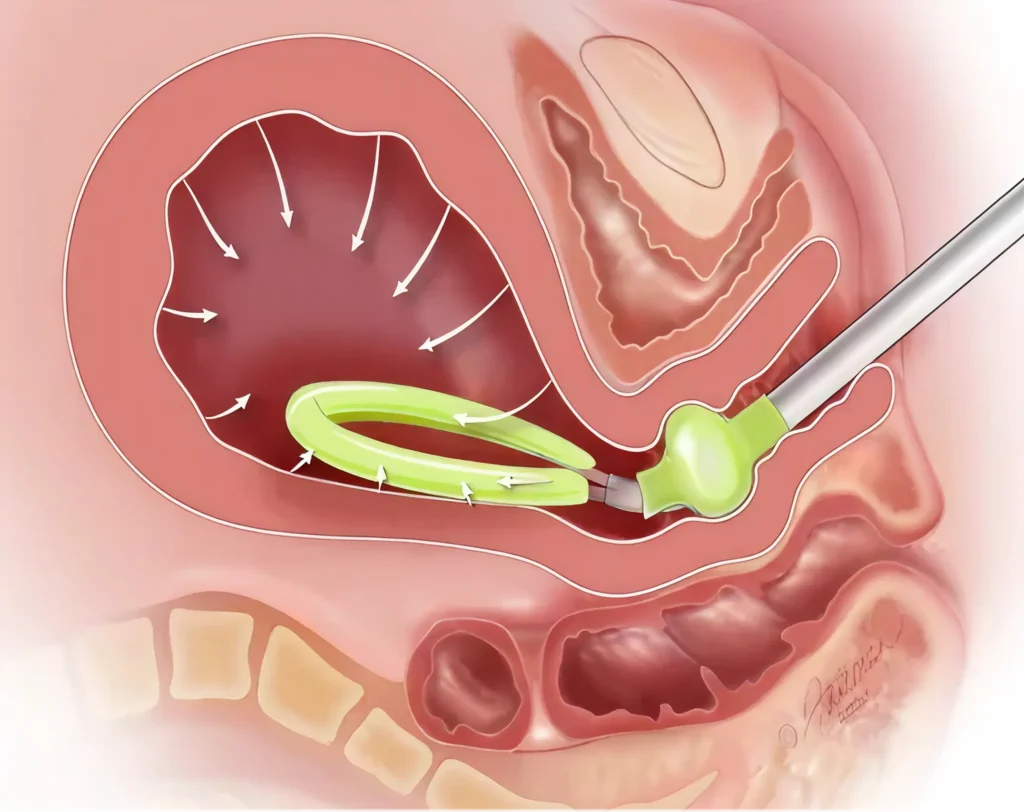
When connected to a low-level vacuum source (80 mm Hg +/- 10 mm Hg; not to exceed 90 mm Hg), JADA establishes a vacuum within the uterus, causing the uterus to contract and leading to control of the bleeding. In a clinical study (n=107), contraction of the uterus was observed in a median of 1 minute with control of bleeding observed in a median of 3 minutes.1


The PEARLE study was a prospective, multicenter, pivotal, single-arm study designed to assess the safety and effectiveness of the JADA System in treating primary postpartum hemorrhage and abnormal postpartum uterine bleeding, and enrolled 107 subjects.1
Read the publication
The RUBY Study was a multicenter, observational, post-market registry that collected observational data on 800 patients treated with the JADA® System from 16 US sites in the post-market setting.
Read the publication
At controlling postpartum uterine bleeding
In the PEARLE study, 94% of participants met the primary end point: bleeding was controlled successfully with JADA, and no further intervention was required. 1 (n=100/106)

Users said they would recommend JADA
Highly recommended among surveyed clinical investigators, in most cases using JADA for the first time.1 (n=104/107)

In nearly every case, investigators rated JADA easy to use
Highly recommended among surveyed clinical investigators, in most cases using JADA for the first time. 1 (n=105/107)
There were no adverse events deemed definitely related to the device or the procedure, and there was a low rate of possibly device-related adverse events.


JADA is made of silicone and was rated easy to use in a survey. (98%, n=105/107).1

An Intrauterine Loop has interior Vacuum Pores inside a protective Shield to evacuate blood.

An expandable Cervical Seal is filled with sterile fluid to create a seal that holds a vacuum within the uterus.

A connector at the end of the tube is attached to a regulated vacuum source.
| Product Code | Description | Quantity |
|---|---|---|
| 1047196 | JADA System – Vacuum-Induced Hemorrhage Control (JADA-2002) | 3 / box |
94% (n=100) of subjects in the PEARLE Study had bleeding controlled in less than 10 minutes.
Leave JADA in place with vacuum applied until bleeding is controlled for 1 hour and continue to assess if the patient’s uterus is firm, bleeding is controlled, and the patient is stable. Leave JADA in place with vacuum disconnected and fluid removed from the Cervical Seal for a minimum of 30 minutes and continue to check that the bleeding remains controlled, and the uterus remains firm.
Yes. Ensure the cervix is dilated to at least 3 cm before use of JADA and verify that the hysterotomy is closed prior to insertion. JADA should be placed transvaginally; do not place through the incision.
Leave JADA in place with regulated vacuum applied until bleeding is controlled for at least 1 hour, the uterus is firm, and the patient is stable. If patient transfer is necessary, it is up to the healthcare professional’s discretion and clinical judgment.
A portable vacuum can be used as long as it is a regulated vacuum source. It can be set to “continuous” suction, and it can be set to 80 mm Hg (+/- 10 mm Hg).
There is no current restriction around using lubrication with the JADA System.
Yes, if there is no purulent infection.
The tubing that is supplied with the JADA System can hold around 100 mL of blood/fluid.
JADA is indicated for single-patient use. Reinsertion is up to the healthcare professional’s discretion and clinical judgment.
Consider prophylactic antibiotics for prolonged use.

The JADA® System is intended to provide control and treatment of abnormal postpartum uterine bleeding or hemorrhage when conservative management is warranted.
For general inquiries or to connect with a JADA Rep: CustomerCareUSA@Laborie.com
For existing customer support: CustomerCareUSA@Laborie.com
1. D’Alton ME, Rood KM, Smid MC, et al. Intrauterine vacuum-induced hemorrhage-control device for rapid treatment of postpartum hemorrhage. Obstet Gynecol. 2020;136(5):882-891. doi:10.1097/AOG.0000000000004138
2. Goffman DE, Rood KM, Bianco A, et al. Real-World Utilization of an Intrauterine, Vacuum-Induced, Hemorrhage-Control Device. Obstet Gynecol. 2023;142(5):1006-1016. doi:10.1097/AOG.0000000000005366
MKT-01784 [A]